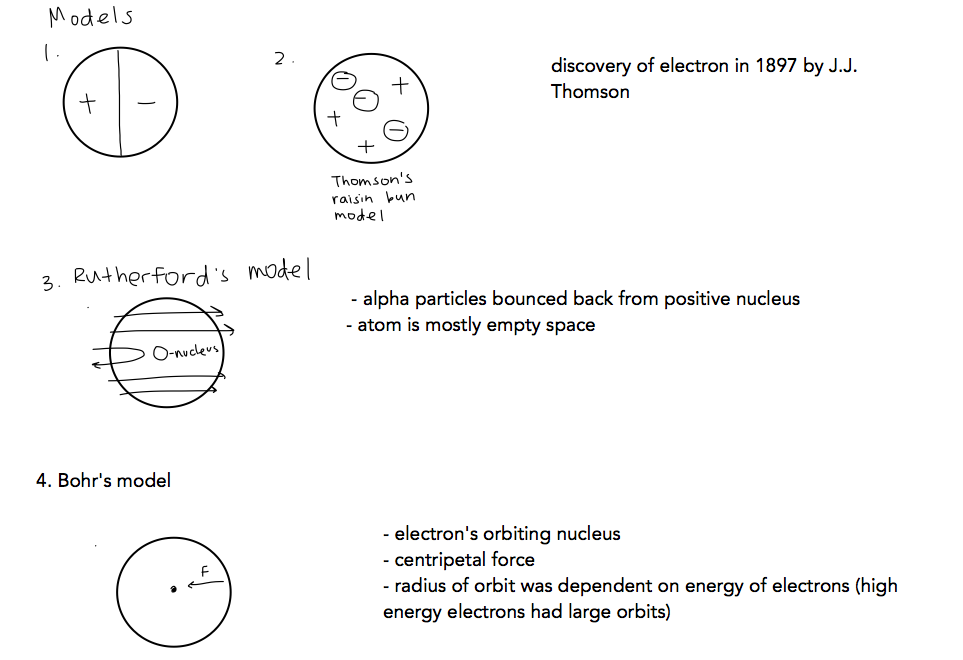
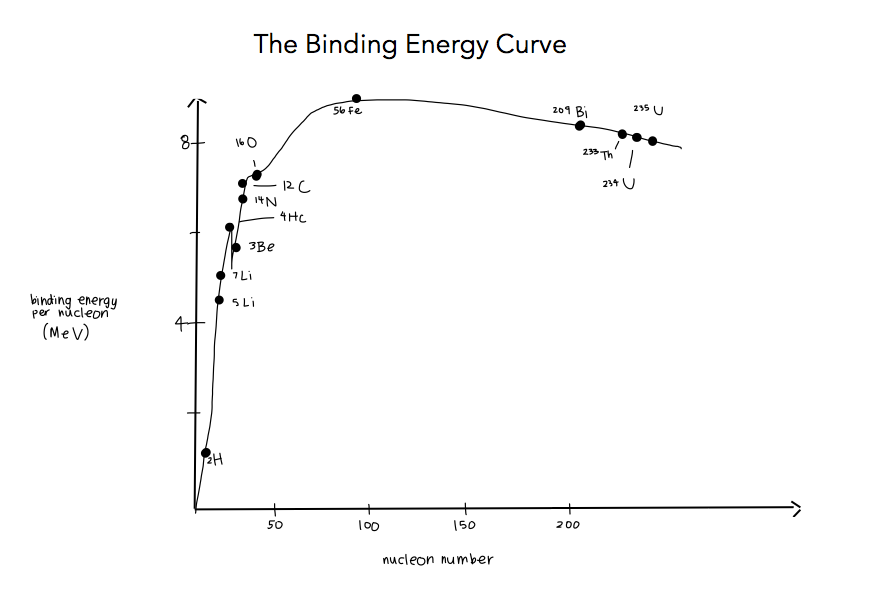
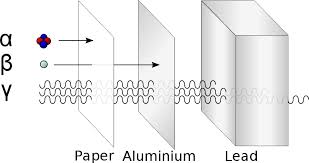
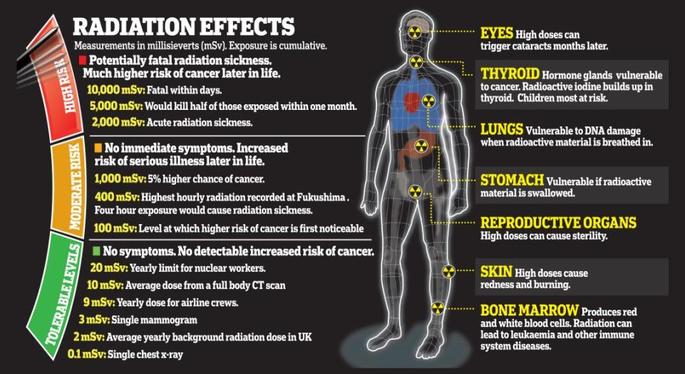
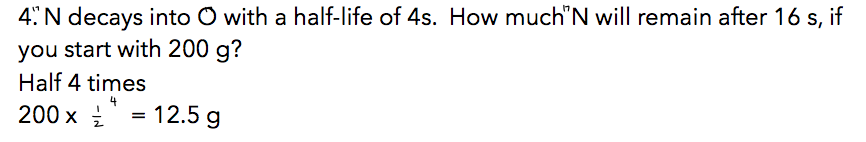7.1 The atom
7.2 nuclear structure
7.3 Radioactive decay
7.4 Half-life
- a nucleus that can decay into another is said to be unstable
- the more energy that is released the more unstable it is
Rate of decay is proportional to the number of nuclei.
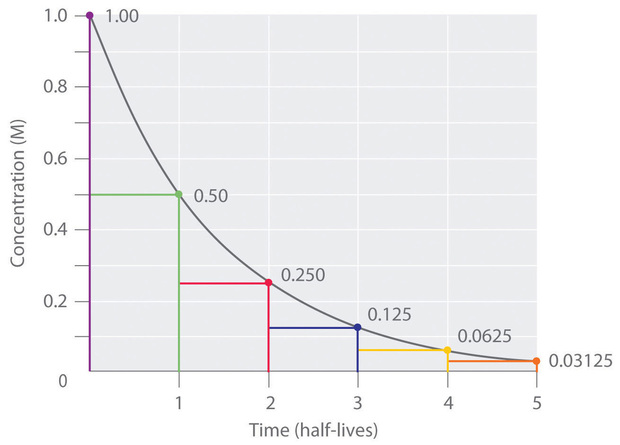
Half-life = time taken for half of the nuclei in a sample to decay
Example:
- the more energy that is released the more unstable it is
Rate of decay is proportional to the number of nuclei.
Half-life = time taken for half of the nuclei in a sample to decay
Example:
Sample of an exponential decay curve:
7.5 nuclear reactions
Nuclear Transmutation: the changing of one chemical element into another. Since it involves a change in the nucleus of an atom, it is a nuclear reaction. Transmutation is the process of changing the number of protons, which identifies its chemical element. When the total protons are changed, the atom is transmuted into atom of another element.
Natural transmutation is responsible for the creation of all the chemical elements we observe naturally. Most of this happened in the distant past.
There are many different methods of producing artificial transmutation. Particle accelerators may be used to bombard elements with alpha particles (two protons and two neutrons), deuterons (one proton and one neutron), or nuclei of atoms of elements such as carbon or boron. Some or all of the protons in the bombarding particles may become lodged in the target nucleus, transmuting it into a different element.
Examples of transmutation:
N(14/7) + n(1/0) -> C(14/6) + p(1/1) *nitrogen nucleus absorbs a neutron and gives out a proton, becomes C14
N(14/7) + He(4/2) -> O(17/8) + p(1/1) *nitrogen nucleus reacted with high speed helium nucleus (alpha particle) to form a 17O and a proton
In all nuclear reactions, the neutron number and proton number must balance.
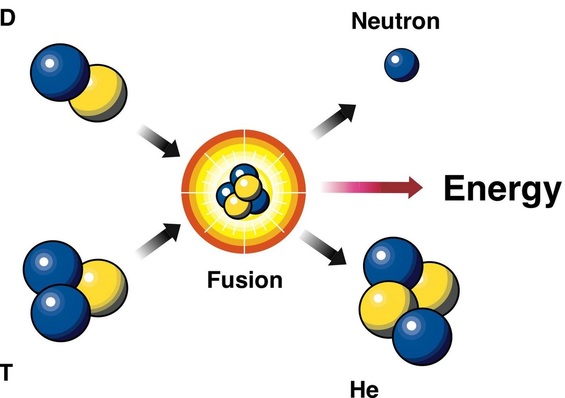
Nuclear Fusion:
-Nuclear: means the process which involves the nucleus
-Fusion: means two or more entities coming together or merging together to form one single entity.
-Nuclear fission: Fusion or combination of nuclei. Nuclear fusion can be defined as a nuclear reaction, in which lighter nuclei are combined together to form heavier product nuclei with the release of enormous amount of energy.
-Mass of heavier nucleus < mass of initial reacting nuclei. Because energy is released during the reaction.
-There are no full time nuclear reactors built so far are fusion reactions, as it is very hard to control such an enormous energy release.
Nuclear Fission
Additional info: Nuclear fission produces energy for nuclear power and to drive explosion of nuclear weapons.
Natural transmutation is responsible for the creation of all the chemical elements we observe naturally. Most of this happened in the distant past.
There are many different methods of producing artificial transmutation. Particle accelerators may be used to bombard elements with alpha particles (two protons and two neutrons), deuterons (one proton and one neutron), or nuclei of atoms of elements such as carbon or boron. Some or all of the protons in the bombarding particles may become lodged in the target nucleus, transmuting it into a different element.
Examples of transmutation:
N(14/7) + n(1/0) -> C(14/6) + p(1/1) *nitrogen nucleus absorbs a neutron and gives out a proton, becomes C14
N(14/7) + He(4/2) -> O(17/8) + p(1/1) *nitrogen nucleus reacted with high speed helium nucleus (alpha particle) to form a 17O and a proton
In all nuclear reactions, the neutron number and proton number must balance.
Nuclear Fusion:
-Nuclear: means the process which involves the nucleus
-Fusion: means two or more entities coming together or merging together to form one single entity.
-Nuclear fission: Fusion or combination of nuclei. Nuclear fusion can be defined as a nuclear reaction, in which lighter nuclei are combined together to form heavier product nuclei with the release of enormous amount of energy.
-Mass of heavier nucleus < mass of initial reacting nuclei. Because energy is released during the reaction.
-There are no full time nuclear reactors built so far are fusion reactions, as it is very hard to control such an enormous energy release.
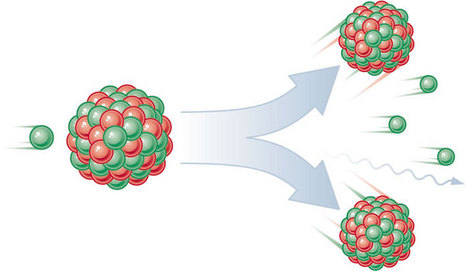
Nuclear Fission
- Nuclear fission is a process in nuclear physics in which the nucleus of an atom splits into two or more smaller nuclei as fission products, and usually some by-product particles.
- In other words, nuclear fission is another form of elemental transmutation.
- The by-products include free neutrons, photons usually in the form gamma rays, and other nuclear fragments such as beta particles and alpha particles.
- Fission of heavy elements is an exothermic reaction and can release substantial amounts of useful energy both as gamma rays and as kinetic energy of the fragments.
Additional info: Nuclear fission produces energy for nuclear power and to drive explosion of nuclear weapons.