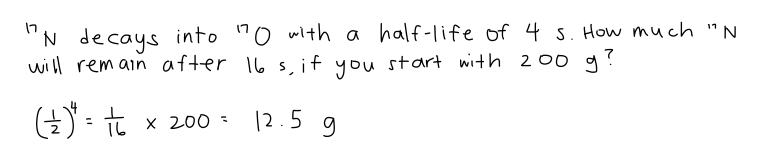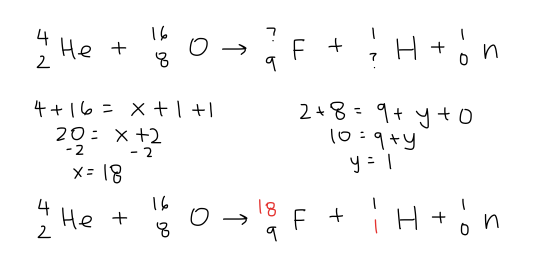Assessment statements
7.1 The atom
7.1.1 Describe a model of the atom that features a small nucleus surrounded by electrons.
The Rutherford model states that the atom features a small nucleus surrounded by electrons. The atom is mostly empty space.
The Rutherford model states that the atom features a small nucleus surrounded by electrons. The atom is mostly empty space.
7.1.2 Outline the evidence that supports a nuclear model of the atom
Some of the alpha particles bounced back, which means there must be a very small particle in the atom. It also had to be positive because it pushed the positive alpha particles away.
Some of the alpha particles bounced back, which means there must be a very small particle in the atom. It also had to be positive because it pushed the positive alpha particles away.
7.1.3 Outline one limitation of the simple model of the nuclear atom.
It was found that if light from hydrogen atoms is analyzed, it was made up of all the frequencies predicted by the model. This model only worked for hydrogen.
Another problem was that accelerating charges give out electromagnetic radiation and lose energy. If electrons lose energy, then they would spiral into the nucleus but this does not happen.
It was found that if light from hydrogen atoms is analyzed, it was made up of all the frequencies predicted by the model. This model only worked for hydrogen.
Another problem was that accelerating charges give out electromagnetic radiation and lose energy. If electrons lose energy, then they would spiral into the nucleus but this does not happen.
7.1.4 Outline evidence for the existence of atomic energy levels.
A change in energy level results in the emission of light. The energy of a wave was believed to be related to its amplitude, not its frequency. According to the law of conservation of energy, each photon has an energy hf (h = planck's constant, 6.6 x 10^/34)
A change in energy level results in the emission of light. The energy of a wave was believed to be related to its amplitude, not its frequency. According to the law of conservation of energy, each photon has an energy hf (h = planck's constant, 6.6 x 10^/34)
7.2 nuclear structure
7.1.5 Explain the terms nuclide, isotope, and nucleon.
Nuclide is a combination of protons and neutrons that make up a nucleus. Isotopes are nuclei with the same number of protons but different numbers of neutrons. Nucleon are the particles of the nucleus including protons and neutrons.
Nuclide is a combination of protons and neutrons that make up a nucleus. Isotopes are nuclei with the same number of protons but different numbers of neutrons. Nucleon are the particles of the nucleus including protons and neutrons.
7.1.6 Define nucleon number A.
The number of protons and neutrons in the nucleus.
The number of protons and neutrons in the nucleus.
7.1.7 Describe the interactions in a nucleus.
There are two types of particles in the nucleus, protons and neutrons.
There are two types of particles in the nucleus, protons and neutrons.
7.3.3 Define the term unified atomic mass unit.
1/12 of the mass of a carbon-12 atom. It is approximately equal to the mass of a proton or neutron.
1.660538782(83)×10−27 kg
1/12 of the mass of a carbon-12 atom. It is approximately equal to the mass of a proton or neutron.
1.660538782(83)×10−27 kg
7.3.4 Apply the Einstein mass-energy equivalence relationship.
E = mc^2
E = energy
m = mass
c= 3 x 10^8 (speed of light)
E = mc^2
E = energy
m = mass
c= 3 x 10^8 (speed of light)
7.3.5 Define the concepts of mass defect, binding energy, and binding energy per nucleon.
Mass defect is the difference between the mass of the nucleus and the mass of the separate parts. Binding energy is the amount of work required to pull apart the parts of a nucleus. Binding energy per nucleon is the energy required to separate a proton or neutron from the nucleus.
Mass defect is the difference between the mass of the nucleus and the mass of the separate parts. Binding energy is the amount of work required to pull apart the parts of a nucleus. Binding energy per nucleon is the energy required to separate a proton or neutron from the nucleus.
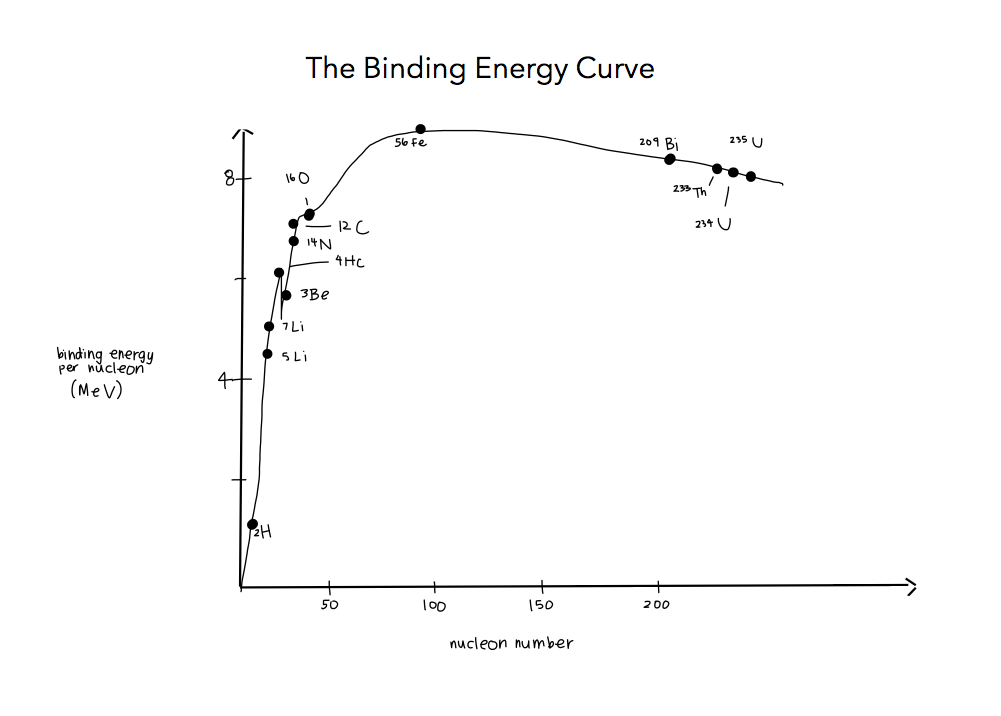
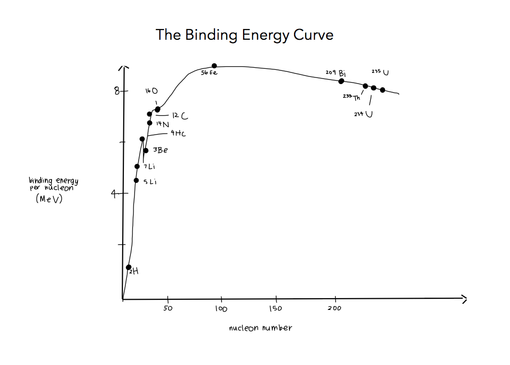
7.3.6 Draw and annotate a graph showing the variation with nucleon number of the binding energy per nucleon.
7.3.7 Solve problems involving mass defect and binding energy.
7.3 radioactive decay
7.2.1 Describe the phenomenon of natural radioactive decay.
If a nucleus emits radiation it can lose energy, but only if the result is an increase in binding energy. This increase and change means that the nucleus has decayed.
If a nucleus emits radiation it can lose energy, but only if the result is an increase in binding energy. This increase and change means that the nucleus has decayed.
7.2.2 Describe the properties of a- and B- particles and y-radiation.
Alpha radiation (a)
- Nucleus loses 2 protons and 2 neutrons
- nucleon number is reduced by 4
- proton number is reduced by 2
Gamma radiation
- no change in particles of the nucleus
- particles just lose energy
- each time a nucleus decays, a photon is emitted
- often emitted when a nucleus is left in an excited state after emitting another form of radiation (alpha or beta)
Alpha radiation (a)
- Nucleus loses 2 protons and 2 neutrons
- nucleon number is reduced by 4
- proton number is reduced by 2
- Beta minus
- - neutron changes to a proton
- - antineutrino is produced
- - loses 1 neutron and gains 1 proton
Gamma radiation
- no change in particles of the nucleus
- particles just lose energy
- each time a nucleus decays, a photon is emitted
- often emitted when a nucleus is left in an excited state after emitting another form of radiation (alpha or beta)
7.2.3 Describe the ionizing properties of a- and B-particles and y-radiation.
Alpha radiation
Beta minus (B-)
Gamma radiation
Alpha radiation
- High ionization
- One alpha particle can ionize 10,000 atoms
- Cannot penetrate paper
- Range of 5 cm in air
- Detection: GM tube, cloud chamber, photographic paper
- it receives almost all of the energy released in the form of kinetic energy
Beta minus (B-)
- Not as heavy as alphas
- Travel with high speed but not as effective at knocking electrons off atoms
- Not as ionizing but produce enough ions to be detected by a GM tube, cloud chamber, or photographic plate
- Pass through more matter and have a longer range in air
- Detection: GM tube, cloud chamber, photographic paper
Gamma radiation
- High energy means that they are absorbed by atomic electrons
- Give the electrons enough energy to leave the atom
7.2.4 Outline the biological effects of ionizing radiation.
When radiation ionizes atoms that are part of a living cell it can hinder the cell's ability or rupture the cell wall. The effect on cells part of a vital organ can even lead to death, but in minor cases the result is a burn. The amount of harm that radiation can cause depends on the number and energy of the particles. A very high dose can affect the central nervous system in 2-3 days, leading to loss of coordination and/or death. A medium dose can damage the stomach and intestine in weeks, leading to sickness, diarrhea, and possibly death. A low dose results in a loss of hair, bleeding, and diarrhea. With a "safe" dose the risk is the smallest and are outweighed by the benefits. After long term exposure to radiation, the probability of getting cancer or having a child with deformities increases.
When radiation ionizes atoms that are part of a living cell it can hinder the cell's ability or rupture the cell wall. The effect on cells part of a vital organ can even lead to death, but in minor cases the result is a burn. The amount of harm that radiation can cause depends on the number and energy of the particles. A very high dose can affect the central nervous system in 2-3 days, leading to loss of coordination and/or death. A medium dose can damage the stomach and intestine in weeks, leading to sickness, diarrhea, and possibly death. A low dose results in a loss of hair, bleeding, and diarrhea. With a "safe" dose the risk is the smallest and are outweighed by the benefits. After long term exposure to radiation, the probability of getting cancer or having a child with deformities increases.
7.2.5 Explain why some nuclei are stable while others are unstable.
Iron is the most stable. The electric repulsion tends to push them apart in nuclei where there is a lot of protons. This means that large nuclei are less stable.
Iron is the most stable. The electric repulsion tends to push them apart in nuclei where there is a lot of protons. This means that large nuclei are less stable.
7.4 half-life
7.2.6 State that radioactive decay is a random and spontaneous process and that the rate of decay decreases exponentially with time. Radioactive decay is a random and spontaneous process and that the rate of decay decreases exponentially with time.
7.2.7 Define the term radioactive half-life.
It is the time taken for half of the nuclei in a sample to decay.
It is the time taken for half of the nuclei in a sample to decay.
7.2.8 Determine the half-life of a nuclide from a decay curve.
You determine how much time it takes for the radioactivity to fall by half.
You determine how much time it takes for the radioactivity to fall by half.
7.2.9 Solve radioactive decay problems involving integral numbers of half-lives.
7.5 nuclear reactions
7.3.1 Describe and give an example of an artificial (induced) transmutation.
The process that a nuclei is changed form one form to another by emitting radiative particles by human is an artificial transmutation. An example is a nitrogen can be transformed into hydrogen by combining its nucleus with an alpha particle.
The process that a nuclei is changed form one form to another by emitting radiative particles by human is an artificial transmutation. An example is a nitrogen can be transformed into hydrogen by combining its nucleus with an alpha particle.
7.3.2 Construct and complete nuclear equations.
7.3.8 Describe the processes of nuclear fission and nuclear fusion.
Nuclear fission is if one large nucleus is split into two smaller ones. Nuclear fusion is the joining up of two small nuclei to form one big one.
Nuclear fission is if one large nucleus is split into two smaller ones. Nuclear fusion is the joining up of two small nuclei to form one big one.
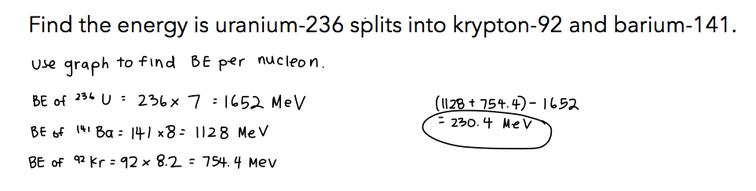
7.3.9 Apply the graph in 7.3.6 to account for the energy release in the processes of fission and fusion.
7.3.10 State that nuclear fusion is the main source of the sun's energy.
Nuclear fusion is the main source of the sun's energy.
Nuclear fusion is the main source of the sun's energy.
7.3.11 Solve problems involving fission and fusion reactions.